- Plot No 459, JLPL, Sector 82, Mohali Sas Nagar, Punjab- 140308
- Info@kronecare.com
Kronebiocare is a trusted name in nutraceutical third party manufacturing services, delivering reliable contract manufacturing and private label solutions for growing supplement brands. Based in India, we support businesses with high-quality production standards, stable formulations, and efficient turnaround timelines.
Our nutraceutical manufacturing services are designed for startups, distributors, and established companies looking to outsource production without compromising on quality. From formulation development to final packaging, we provide complete manufacturing support under one roof.
With a focus on compliance, transparency, and scalable production capacity, we help brands confidently launch and expand their nutraceutical product range in competitive markets.
Our nutraceutical third party manufacturing services in India are designed to help brands outsource production with complete confidence. We manage the entire manufacturing cycle including raw material sourcing, formulation standardization, blending, processing, packaging, labeling, and final dispatch.
Our facility operates under strict quality control systems to ensure every batch meets safety and stability standards. Whether you are launching a new supplement brand or expanding your existing portfolio, we provide scalable manufacturing solutions with transparent communication and reliable timelines.
We provide comprehensive nutraceutical contract manufacturing services tailored to your brand requirements. From product concept discussion and formulation development to commercial-scale production, our team ensures precision, compliance, and consistency at every stage.
Our contract manufacturing model allows businesses to focus on marketing and distribution while we handle production efficiency, quality testing, documentation, and packaging execution. This reduces operational burden and ensures cost-effective, professional manufacturing support.
Our private label nutraceutical manufacturing services allow you to launch your own supplement brand without investing in infrastructure.
We provide customized formulations, branding coordination, packaging solutions, and complete manufacturing support.
We specialize in custom nutraceutical manufacturing services tailored to your specific formulation requirements. Whether you need new product development or modification of existing formulas, our team ensures quality, safety, and compliance.
To support new businesses, we offer flexible and low MOQ nutraceutical manufacturing services. This helps startups enter the market with controlled investment while maintaining premium product standards.
As an experienced nutraceutical export manufacturing company, we provide manufacturing solutions aligned with international quality standards. We assist with documentation and packaging requirements for global markets.














Third party contract manufacturing has become a strategic solution for nutraceutical and pharmaceutical brands looking to scale efficiently without investing in production infrastructure. With growing global demand for health supplements, outsourcing manufacturing ensures cost control, regulatory compliance, and faster market entry.
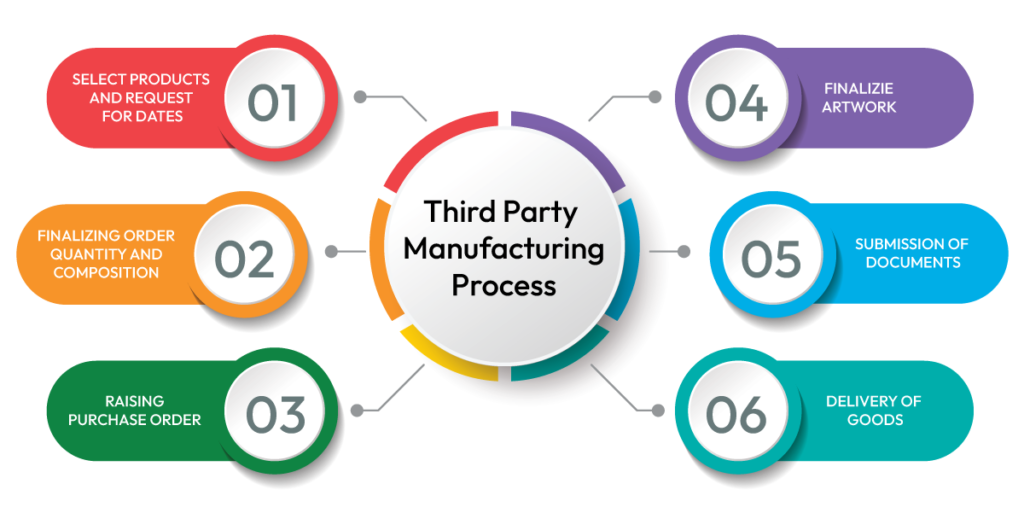
A well-structured manufacturing process ensures transparency, quality assurance, and timely delivery at every stage. Below is a step-by-step overview of how our third party contract manufacturing process works.
Requirement analysis is the foundation of a successful third party contract manufacturing process. This stage ensures that both the brand owner and the manufacturer are aligned on product expectations, regulatory standards, and commercial objectives before production begins. In nutraceutical third party manufacturing, clarity at the initial stage prevents costly errors, delays, and reformulation issues later in the cycle.
During requirement analysis, key factors such as dosage form selection (capsules, tablets, syrups, sachets, softgels, powders), ingredient composition, strength, target health segment, and packaging preferences are discussed in detail. The manufacturer evaluates technical feasibility, stability considerations, and sourcing requirements for raw materials. This ensures the final formulation is safe, compliant, and commercially viable.
In addition, market positioning and pricing strategy are considered to determine minimum order quantity (MOQ) and batch size planning. For startups and growing brands, this step is critical in balancing investment control with scalable production capacity.
A structured requirement analysis process in third party contract manufacturing builds transparency, establishes realistic timelines, and sets clear quality benchmarks. It acts as the blueprint that guides formulation development, procurement planning, documentation preparation, and final manufacturing execution.
Batch production is the operational core of third party contract manufacturing. Once formulation specifications and documentation are approved, manufacturing begins under controlled and standardized conditions. Every stage of batch processing follows predefined standard operating procedures to maintain consistency, safety, and regulatory compliance.
In nutraceutical third party manufacturing, quality control begins with raw material inspection. Ingredients are verified for identity, purity, and conformity to approved specifications before entering the production line. During processing, in-process quality checks are conducted to monitor parameters such as weight variation, blend uniformity, moisture levels, and dosage accuracy.
Quality assurance teams oversee each stage to ensure that capsules, tablets, powders, or liquid formulations meet defined standards. After manufacturing, finished goods undergo final quality testing, which may include microbial analysis, dissolution testing, and physical verification depending on product type.
Comprehensive documentation is maintained for batch traceability, audit readiness, and regulatory compliance. This structured quality management system strengthens reliability in third party contract manufacturing and protects brand reputation in competitive nutraceutical markets.
Consistent batch production supported by strict quality control ensures long-term product stability, consumer safety, and regulatory alignment.
Packaging and dispatch represent the final execution stage in the third party contract manufacturing process. Once batch production and quality approval are completed, products move to packaging as per finalized artwork and labeling specifications. Proper packaging is essential not only for product protection but also for regulatory compliance and brand presentation.
In nutraceutical third party manufacturing, packaging includes accurate label placement, batch coding, manufacturing date, expiry date, and ingredient declaration as per compliance requirements. Packaging materials are inspected before use to ensure durability and correctness. This minimizes the risk of mislabeling or transportation damage.
After packaging, finished goods are counted, sealed, and prepared for shipment according to agreed logistics plans. Dispatch timelines are aligned with commercial commitments discussed during order finalization. Secure handling and transport arrangements ensure safe delivery to warehouses, distributors, or export destinations.
Documentation such as batch records, invoices, and compliance certificates are prepared to support smooth transit and regulatory verification if required. Efficient packaging and dispatch management reflect operational discipline in third party contract manufacturing services.
Timely delivery strengthens client confidence and ensures consistent product availability in the market.
Choosing the right nutraceutical third party manufacturing company directly impacts product quality, regulatory compliance, and long-term brand growth. A serious buyer should evaluate manufacturing capability, dosage form availability, quality control systems, documentation standards, and production scalability before finalizing a contract manufacturing partner.
A reliable nutraceutical contract manufacturer should offer multiple dosage formats such as capsules, tablets, softgel capsules, protein powders, sachets, syrups, gummies, and drops. This ensures flexibility in launching products across different health categories like immunity support, bone and joint care, heart health, liver support, sports nutrition, women’s wellness, and diabetes management.
Manufacturing approach is equally important. The company should follow structured batch production processes, raw material verification protocols, in-process quality checks, and final product testing before dispatch. Documentation transparency and traceability systems are critical for long-term compliance and export readiness.
From a buyer’s intent perspective, scalability matters. A good third party nutraceutical manufacturer should support both low MOQ production for startups and bulk manufacturing for established brands. Timely delivery, cost clarity, and stable formulations are strong indicators of operational discipline.
Selecting a manufacturing partner is not just about price — it is about consistency, compliance, and capacity to grow with your brand.
Nutraceutical contract manufacturing allows brands to focus on marketing, distribution, and product positioning while outsourcing the complexities of production. This model reduces capital investment in infrastructure, machinery, regulatory approvals, and skilled workforce management.
Under nutraceutical third party manufacturing services, production is handled by a specialized facility that manages formulation development, raw material procurement, batch manufacturing, quality assurance, packaging, and dispatch. This structured manufacturing approach ensures that supplements such as capsules, tablets, protein powders, softgels, and syrups are produced consistently under defined quality standards.
From a medical segment perspective, contract manufacturing enables brands to expand across multiple therapeutic categories without operational stress. Companies can launch immunity boosters, joint care supplements, heart health formulations, digestive support products, or sports nutrition lines using an established manufacturing setup.
Buyer intent often revolves around cost control and scalability. Nutraceutical manufacturing services offer predictable production timelines, defined batch sizes, and optimized cost structures. This helps businesses manage inventory efficiently and respond to market demand without production bottlenecks.
For startups, it lowers entry barriers. For growing brands, it improves operational efficiency. For large distributors, it ensures stable supply chains. That is why nutraceutical third party manufacturing has become a preferred strategy in the supplement industry.
Third party nutraceutical manufacturing covers a wide spectrum of dosage forms and therapeutic segments. A professional nutraceutical manufacturing facility typically supports capsules, tablets, softgel capsules, protein powders, sachets, syrups, effervescent tablets, granules, gummies, and oral drops.
These dosage forms serve multiple health and medical categories including immunity support, bone and joint health, heart care, liver detox support, kidney wellness, blood sugar management, brain and memory enhancement, sports nutrition, digestive health, and general multivitamin supplementation.
From a formulation perspective, products may include herbal extracts, vitamins, minerals, amino acids, probiotics, antioxidants, protein blends, and specialized nutraceutical combinations. Custom formulation options allow brands to create unique market positioning rather than relying on generic compositions.
Buyer intent often varies depending on market positioning. Some clients look for low MOQ nutraceutical manufacturing to test product demand. Others require bulk contract manufacturing for national distribution or export supply. A capable third party manufacturer must support both scenarios while maintaining consistent quality and batch stability.
The advantage of outsourcing nutraceutical production is operational flexibility. Brands can introduce new categories, modify formulations, or scale production volumes without investing in new infrastructure.